By early 2025, over 270 generic drugs were in short supply across the U.S.-a record high. These aren’t rare or experimental medications. They’re the pills and injections millions of Americans rely on every day: antibiotics, chemotherapy drugs, IV fluids, epinephrine, and insulin. When these drugs vanish from pharmacy shelves, it’s not just an inconvenience. It’s a crisis that delays cancer treatments, forces doctors to use riskier alternatives, and puts lives at risk.
Why Generic Drugs Are the First to Go Missing
Generic drugs make up 90% of all prescriptions filled in the U.S., but they account for just 13% of total drug spending. That’s because they’re cheap-sometimes under $5 per dose. And that’s exactly the problem.Manufacturers of brand-name drugs can afford to stockpile inventory, invest in multiple production sites, and absorb cost spikes. Generic drug makers? They operate on razor-thin margins. A single factory shutdown, a delayed shipment, or a quality inspection failure can wipe out their entire profit-and push them out of the market entirely.
Take sterile injectables, like IV antibiotics or chemotherapy drugs. These require clean rooms, specialized equipment, and highly trained staff. It’s expensive and slow to make. But because these drugs sell for pennies, manufacturers have consolidated production into just one or two facilities worldwide. One tornado in 2023 destroyed a Pfizer plant and knocked out 15 critical medications. A quality violation in India shut down cisplatin production, a key cancer drug, leaving U.S. hospitals scrambling.
The Global Web of Risk: China, India, and the Missing API
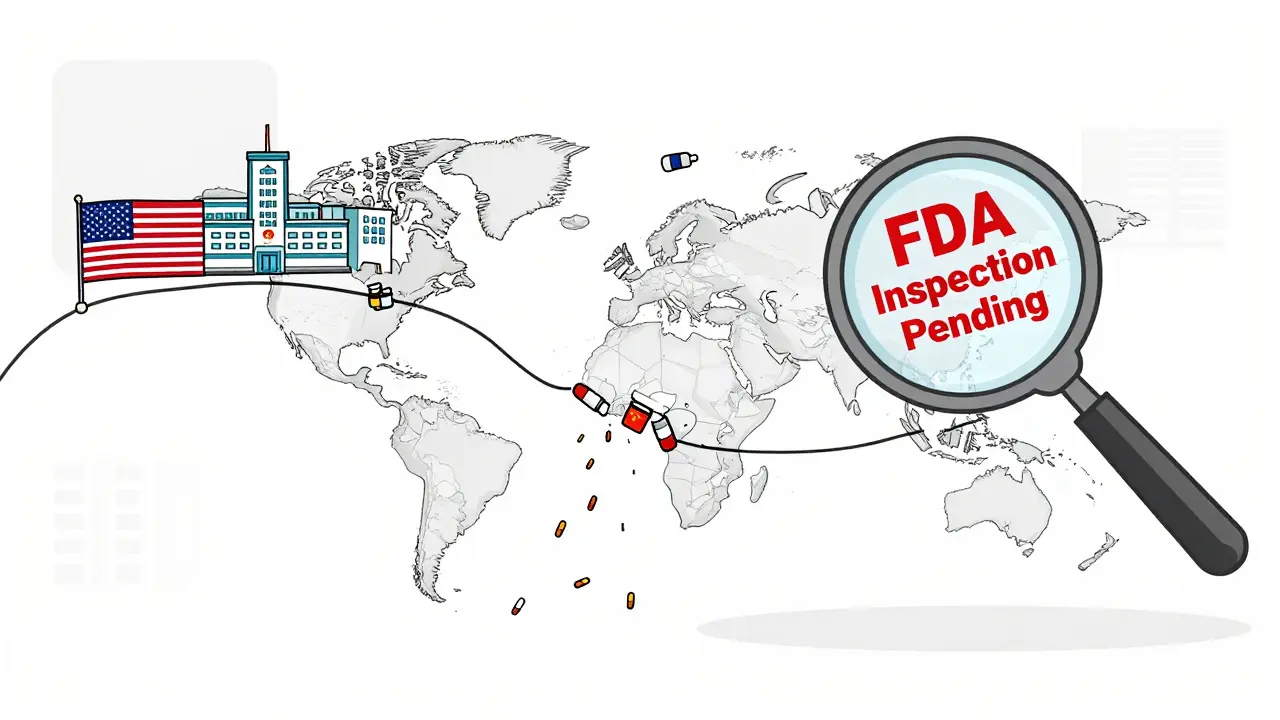
Almost every generic drug starts with an active pharmaceutical ingredient, or API. And here’s the truth: less than 30% of the APIs used in U.S. medicines are made in America. The rest come from just two countries-China and India.China supplies about 40% of the world’s API production. But getting API from China isn’t simple. For years, U.S. regulators flagged reliability issues. Some manufacturers didn’t submit required documentation (Drug Master Files) because they feared inspections or didn’t meet U.S. standards. Even today, the FDA’s ability to inspect these facilities is limited, and many plants operate with outdated equipment.
India handles the next step: turning those APIs into pills and injections. It’s cheaper there, so most generic makers outsource final manufacturing to Indian plants. But when India’s regulatory agencies crack down-or when a factory has a contamination issue-dozens of drugs vanish overnight. There’s no backup. No redundancy. Just one link in a very long chain.
Why Onshoring Won’t Fix This Overnight
You’ve probably heard the solution: bring drug manufacturing back to the U.S. It sounds logical. But it’s not realistic.Rebuilding domestic capacity for sterile injectables alone would cost $20 to $30 billion and take 5 to 7 years. You’d need new factories, certified engineers, trained technicians, and a supply chain for raw materials-all while competing with countries that pay workers a fraction of U.S. wages. And even if you did, who would buy the drugs? Not hospitals. Not insurers. Not patients. Because the price would have to rise just to cover costs-and that’s not what the market allows.
Some lawmakers push tariffs on imported APIs to force production back home. But experts warn this could backfire. A 50% to 200% tariff would spike prices for drugs like heparin, epinephrine, and doxycycline. Hospitals would pay more. Patients would pay more. And manufacturers might just quit making the cheapest drugs altogether, making shortages worse.
Who Pays the Real Price?
It’s not just hospitals or pharmacies. It’s patients.One hospital pharmacist told a survey they spend 20 to 30% of their workweek managing shortages. That means tracking down alternatives, compounding drugs from scratch, or calling other hospitals to borrow stock. Nurses have to switch patients to less effective or more dangerous drugs. Oncologists delay treatments. Emergency rooms run out of epinephrine for allergic reactions.
In 2024, the number of active drug shortages hit 323-the highest ever. That’s more than the peak during the pandemic. And it’s not getting better. The FDA’s shortage list is always changing. One day, a drug is back in stock. The next, it’s gone again. No warning. No explanation. Just silence.

What’s Being Done-and Why It’s Not Enough
There are proposals. The American Hospital Association wants a national stockpile of 6 months’ worth of critical generics. Congress has introduced bills to require manufacturers to report potential shortages six months in advance. Some want mandatory labeling so you know if your drug’s API came from China or India.But implementation is slow. Federal agencies like the FDA and HHS have seen staff cuts and budget reductions. Inspections are falling behind. Regulatory processes are stuck in bureaucracy. Even when a solution is agreed on, it takes years to move from paper to practice.
Meanwhile, the market keeps pushing prices down. Generic drug buyers-hospitals, pharmacies, Medicaid-choose the cheapest option every time. That rewards the lowest bidder, not the most reliable one. So manufacturers compete on price, not resilience. And the system breaks.
The Path Forward: It’s Not Just About Making More
Fixing this won’t mean building 10 new factories in Ohio. It means changing how we value these drugs.We need to pay a little more for the essentials-especially sterile injectables and older generics that no one wants to make. That money should go toward building backup capacity, investing in quality control, and diversifying suppliers. Not just in China and India, but in places like Mexico, Poland, or even the U.S., if they can meet standards.
We need transparency. Patients and providers deserve to know where their drugs come from. Labels should say: “API sourced from China.” That’s not fearmongering. It’s accountability.
And we need incentives. Right now, there’s no reward for being a reliable manufacturer. If you make 100,000 doses of a $2 antibiotic and never miss a shipment, you get nothing. But if you cut corners, save $10,000, and get away with it? You win. That’s not a market. It’s a trap.
The system isn’t broken because of the pandemic. It was broken long before. And unless we fix the economics-not just the logistics-we’ll keep seeing the same shortages, year after year, with the same dangerous consequences.


8 Comments
This is what happens when you treat life-saving medicine like a commodity instead of a human right. We’ve turned healthcare into a casino, and the poor are always betting with their last dollar. Every time a nurse has to dig through drawers for a substitute drug, someone’s life is on the line-and nobody’s even flinching. We’re not just broken, we’re numb.
It is, indeed, a systemic failure-one that stems from a fundamental misalignment between market incentives and public health imperatives. The pricing structure for generic pharmaceuticals is not merely inefficient; it is ethically indefensible. When profit margins are so thin that manufacturers cannot afford redundancy, quality control, or even basic contingency planning, the system is not just flawed-it is actively dangerous.
Moreover, the reliance on geopolitical hotspots for active pharmaceutical ingredients introduces an unacceptable level of risk. It is not merely a supply chain issue; it is a national security concern.
THEY KNOW WHAT THEY’RE DOING!!! THE BIG PHARMA CORPORATIONS AND THE GOVERNMENT ARE IN BED TOGETHER-THEY WANT YOU SICK SO THEY CAN KEEP SELLING YOU STUFF!!! WHY DO YOU THINK THEY LET CHINA MAKE 40% OF OUR DRUGS?!? IT’S A TRAP!!! THEY’RE LETTING THE SYSTEM COLLAPSE SO THEY CAN PUSH “PREMIUM” DRUGS AT 10X THE PRICE!!!
THE FDA IS CORRUPT!!! THE INSPECTIONS ARE A JOKE!!! THEY’RE ALL IN ON IT!!!
As someone from India, I see this every day. Our factories aren’t lazy or careless-we’re doing our best with limited resources. But when U.S. buyers demand $0.10 per pill and then inspect like they’re hunting for terrorists? It’s impossible. We’re not the villains here. We’re the ones holding the chain together while everyone else screams for cheaper prices.
Instead of blaming us, why not help us upgrade? Fund better labs. Pay a fair price. Let us be part of the solution-not the scapegoat.
My cousin is on chemo and they switched her to a different generic last month. She got sick for two weeks because the new one didn’t absorb right. No one told her why. No one apologized. Just silence. This isn’t policy-it’s personal. We need to stop pretending this is just a “supply issue.” It’s a failure of care.
While the systemic issues outlined are profound, one often-overlooked variable is the role of formulary management within hospital pharmacy and therapeutics committees. These committees, under pressure to reduce costs, routinely select the lowest-bidder generic without evaluating manufacturing reliability, historical supply stability, or bioequivalence variance. A more nuanced evaluation framework-incorporating not only price but also supplier performance metrics-could mitigate many shortages before they occur.
Additionally, the FDA’s Drug Shortage Program requires enhanced data integration from manufacturers. Real-time inventory reporting, coupled with predictive analytics, could enable proactive redistribution rather than reactive crisis management.
Just read this whole thing and honestly? I didn’t realize how fragile our meds are. I always just assumed the pharmacy had what I needed. Now I’m scared to even fill a prescription. Maybe we need to start treating these drugs like water or electricity-basic infrastructure. Not a product to be bid on like a garage sale.
It’s all a hoax. The shortages are staged. The FDA and Big Pharma are hiding the real drugs-ones that actually work-and forcing us to take the cheap ones that cause side effects. They’re using this to push vaccines and biologics. You think epinephrine is hard to find? Wait till they replace it with nanotech serums. They’re already testing them in your water.