
Explore the key differences between retail and hospital pharmacy substitution practices, from legal frameworks to safety protocols.

Explore the key differences between retail and hospital pharmacy substitution practices, from legal frameworks to safety protocols.

Foreign-made generic drugs supply 90% of U.S. prescriptions. With new FDA rules in 2026, unannounced inspections are now the norm. Here’s how oversight is changing-and what it means for your medicine.

When your prescription switches to a generic drug, communication with your pharmacy can prevent side effects and ensure your treatment stays effective. Learn what to ask, when to speak up, and how to stay in control.
Bioequivalence testing for combination products like fixed-dose combos and drug-device systems is far more complex than for single drugs. Regulatory hurdles, inconsistent methods, and high costs delay generic access - but new modeling tools are starting to change that.

Discover how generic medicines travel from raw ingredients to your local pharmacy. Learn about manufacturing, distribution, pricing challenges, and key stakeholders in the supply chain.
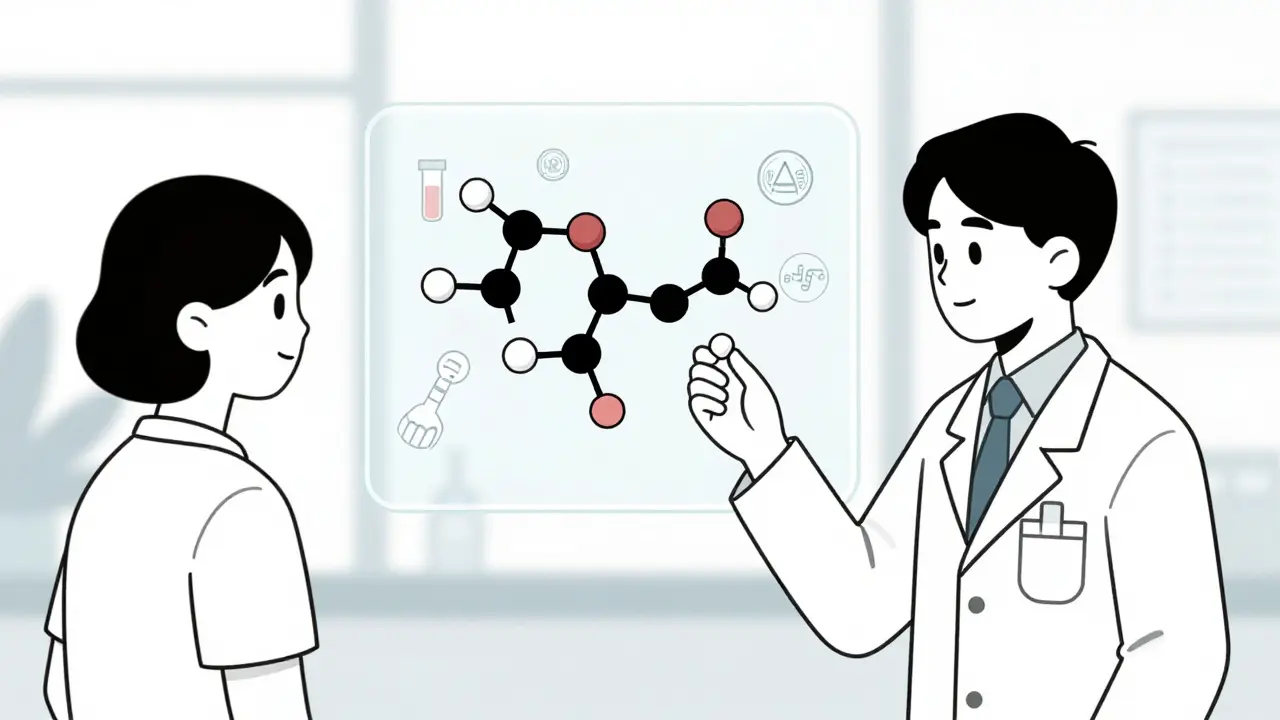
Generic drugs must meet the same FDA standards as brand-name drugs, including pharmaceutical and bioequivalence testing. Learn how the FDA ensures quality, safety, and effectiveness through rigorous manufacturing and testing protocols.

Learn how to talk to your doctor about generic medications-why they’re just as safe and effective as brand-name drugs, how much you can save, and what to say if you’re unsure.
Bioequivalence testing ensures generic drugs deliver the same active ingredient at the same rate as brand-name versions, making them safe and effective. This science protects patients while cutting healthcare costs.

Medicaid saves billions by using generic drugs, cutting prescription costs for low-income patients from $56 to just $6 per fill. Learn how the system works, where savings are lost, and how patients can get the most out of their coverage.
Generic drugs are safe when made under strict quality controls that prevent counterfeits. Learn how cGMP, track-and-trace systems, and advanced testing stop fake medicines before they reach patients.

The FDA ensures generic drugs work just like brand names through strict bioequivalence testing, identical active ingredients, and rigorous manufacturing standards-saving billions without compromising safety or effectiveness.